CDHB
Individualization of patient treatment is the basis of good prescribing. Drug selection and choice of maintenance dose rates are important clinical decisions. Patient characteristics, such as age, weight, presence of renal or liver impairment, diseases, interacting drugs, pregnancy, breast feeding etc. should be considered. See the Pink Book for more detail.
For some drugs with a narrow therapeutic index, drug concentration monitoring is recommended. Laboratory results are reviewed daily and advice provided. Contact the Clinical Pharmacology Registrar for assistance.
For dose individualization of aminoglycosides and vancomycin, contact the ward pharmacist or Drug Information ![]() 80900.
80900.
Drugs (or active metabolites), with a high fraction excreted unchanged (fu) in the urine and a low therapeutic index, require dose-reduction in renal impairment.
Note: eGFR is provided by Canterbury Health Laboratories (using the CKD-EPI formula). Like its predecessor, the MDRD formula, this is unreliable when the creatinine is changing or at extremes of weight.
Calculate CrCl using the modified Cockcroft and Gault equation as follows:

Note: Use actual body weight if this is less than the ideal body weight.
Note: The creatinine must be stable for the estimated renal function to be valid (whether using the eGFR result or the Cockcroft and Gault result). In addition, the further the patient is from normal (height, weight, etc.), the less valid the estimate. For the purpose of the above calculation, normal creatinine clearance is assumed to be 100 mL per minute. Alternatively the estimated glomerular filtration rate (eGFR) supplied by the laboratory can be used for patients whose BMI is close to the normal range.
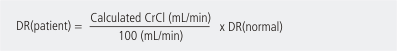
For drugs with fu <0.9, calculate the dose-rate for the patient (DR(patient)) as follows:

Topic Code: 1290